Prophylactic antibiotic regimen for infective endocarditis in dental procedures
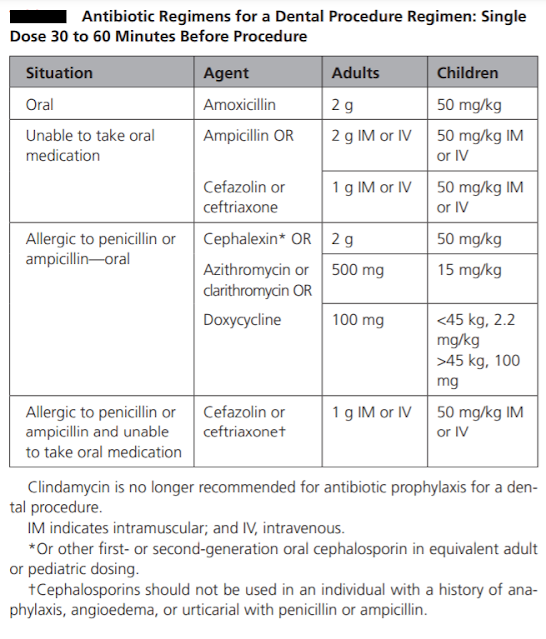
Antibiotic prophylaxis is recommended for invasive dental procedures that involve the manipulation of gingival tissue or periapical region or perforation of the mucosa when performed on high-risk individuals. Australian guidelines have provided a list of dental procedures that are likely to cause a high incidence of bacteraemia that always require prophylaxis. These are as follows: Tooth extraction. Periodontal surgery, subgingival scaling and root planning. Replantation of avulsed teeth. Other surgical procedures such as implant placement or apicoectomy. Procedures that cause a moderate incidence of bacteraemia might be considered for prophylaxis if multiple procedures are being conducted, in cases where the procedure is prolonged, or in the setting of periodontal disease. Antibiotic prophylaxis is not recommended for procedures with a low possibility of bacteraemia such as: Local anaesthetic injections. Dental X-rays. Treatment of superficial caries. Orthodontic appliance placement a...